April 30, 2026
Allocation Notice – $0.91 Pricing Window Closing
Featured Editorial: Krystal Biotech Is Quietly Becoming One of Biotech’s Most Profitable Platforms

|
$RADI $0.91 ▲ |
Pricing Window Closing
Price Changes Tonight at 11:59pm PST. Invest at $0.91/Share.
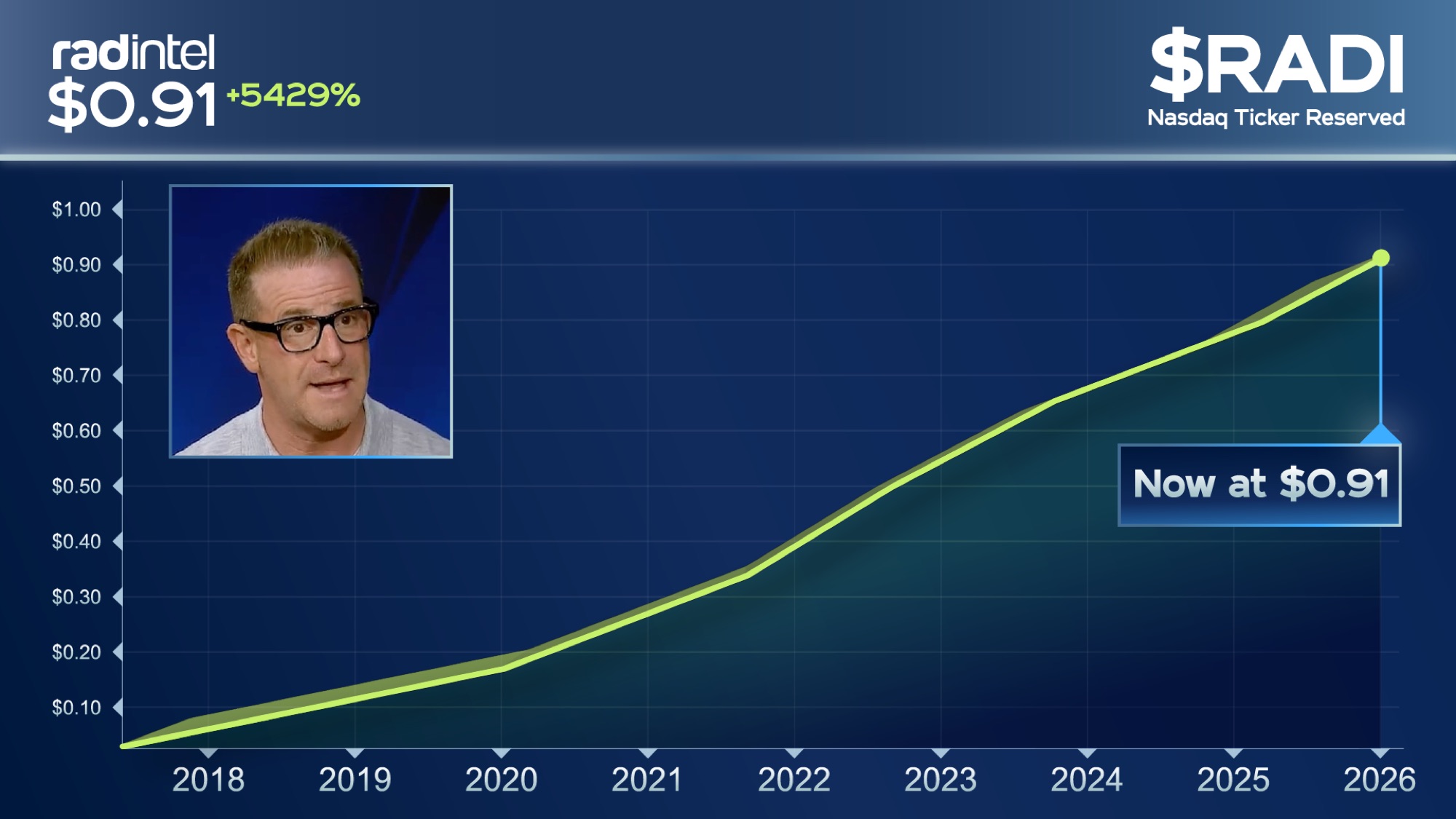
RAD Intel’s current Reg A+ round remains open at $0.91 per share – though that price is scheduled to change. Final window at the current price ends April 30 at 11:59 PM PST.
|
|
|
| ► Invest Now at $0.91/Share |
20,000+ investors already in · $75M+ raised to date
Here’s What $0.91 Per Share Gets You
|
||
|
||
|
||
|
Backed by multiple institutional funds. Selected by the Adobe Design Fund. Supported by early operators from Google, Meta, YouTube, and Amazon.

|
$0.91 pricing window closes April 30 at 11:59 PM PST |
|
|
Lock In $0.91/Share Before the Price Changes. |
|
|
|
|
After tonight at 11:59 PM PST, this entry point is no longer available. |
Featured Editorial
Krystal Biotech Is Quietly Becoming One of Biotech’s Most Profitable Platforms
Most biotech stories are binary. Drug works, stock explodes. Drug fails, stock craters. It’s a coin flip dressed up in clinical trial language.
Krystal Biotech (NASDAQ: KRYS) doesn’t fit that template — and that’s exactly the point.
This is a Pittsburgh-based, commercial-stage genetic medicine company with a product already on the market, already approved in the U.S., Europe, and Japan, and already generating the kind of margins that make software companies look average. VYJUVEK — its flagship therapy for dystrophic epidermolysis bullosa, a devastating rare skin disease — is the first-ever redosable gene therapy cleared by the FDA. Not a one-time infusion. Redosable. That’s a structural distinction that changes the commercial model entirely.
The numbers are hard to ignore. TTM revenue sits at $389 million. Net profit margin is 52.6%. Operating margin is 41.4%. Operating cash flow is roughly $200 million. For a biotech company, those margins don’t look like biotech — they look like enterprise software with FDA exclusivity attached.
Get YOUR Pre-IPO Exposure to ChatGPT’s Trillion Dollar IPO!
No Accreditation… No Qualifications…
What changed the narrative in early 2026 was a cascade of pipeline catalysts that rewired analyst expectations. In January, Krystal announced favorable interim Phase 1 results for KB407 — its inhaled gene therapy targeting cystic fibrosis — with CFTR expression in six out of seven patients. That’s not a small signal. CF is a multi-billion dollar market, and the company is now aligning with the FDA on a registrational Phase 3 study expected to commence by mid-2026. Then in February, the FDA granted Regenerative Medicine Advanced Therapy (RMAT) designation to KB707 for non-small cell lung cancer — a designation designed specifically to accelerate development of promising therapies.
The global expansion story is the part that hasn’t fully priced in yet. VYJUVEK has launched in Germany, France, and Japan, with over 90 patients prescribed across those international markets. The company has secured over 660 reimbursement approvals in the United States alone. Management’s stated goal: at least four marketed rare disease medicines by end of 2030, treating over 10,000 patients worldwide — while remaining profitable throughout.
Analyst conviction has followed. Citigroup raised its price target to $371 in February 2026. TD Cowen went to $306. Guggenheim moved to $284. The consensus sits at Strong Buy. Revenue growth is forecast at approximately 19.8% annually, with earnings expected to grow 22% — both well above broader market projections.
Your Free Options Book Is About to Vanish
In case you missed it… make sure you get your free “Simple Options Trading For Beginners” book before your link expires.
I eventually plan to charge money for this training, so do yourself a favor and download it now…
That way, no matter what it costs in the future, you’ll have a free copy.
Sound good?
The risks are real. VYJUVEK treats a rare disease with a limited patient pool, which caps how fast U.S. revenue can scale. The pipeline candidates — KB407 for CF, KB707 for lung cancer, KB803 for ocular DEB complications — each carry their own clinical and regulatory uncertainty. International reimbursement negotiations in Italy and Japan, expected in H2 2026, are a key catalyst with binary potential. One data readout or reimbursement delay can reset expectations fast.
What’s interesting is the broader setup. Healthcare and biotech have been quietly outperforming in 2026, rotating back into favor after years of underperformance. The sector’s momentum is being backed by real fundamentals — improving earnings visibility, AI-enhanced drug discovery pipelines, and a regulatory environment showing more clarity than it has in years.
Krystal sits at the intersection of a proven commercial product, a platform technology that can generate multiple medicines, and margins that suggest the business model actually works. That combination is rare in biotech. An upcoming earnings update on May 5, 2026 could provide fresh visibility on both U.S. patient dynamics and international expansion timing — two variables that will matter a lot for where this one goes next.